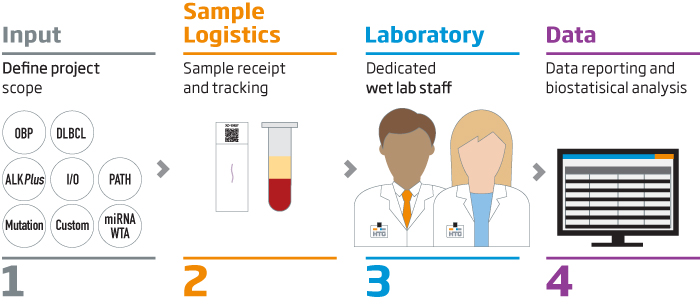
Molecular Profiling and More for Your Samples
VERI/O Laboratory Services supports retrospective molecular profiling studies for biopharma clients and translational medicine researchers. In addition to sample processing services using HTG’s on-market products, we also offer early access to our newest technologies, such as our direct sequencing chemistry that run exclusively in the VERI/O lab.
VERI/O laboratory also provides assay development, sample accessioning, and data analysis services to support studies.
Let VERI/O Laboratory Services be an extension of your organization. Take advantage of the benefits of our
HTG EdgeSeq technology without the up-front capital expense.
VERI/O Laboratory Services:
- Retrospective sample processing
- Pilot studies
- Custom assay development
- Disease-specific validation studies
For custom assays, please contact HTG directly.
Service Offerings
VERI/O Laboratory Services accepts a wide variety of clinical samples for testing, including formalin-fixed, paraffin-embedded (FFPE) tissue, plasma/serum, PAXgene, extracted RNA, and cells. Sample types and amounts are specific to each assay and customer project.
“HTG has been a true partner and collaborator in our drug development program and we truly appreciate their support to meet the regulatory filing deadline for one of our key drug products.”
~ Senior Manager, Clinical Biomarkers—Oncology, Global Pharma Partner
How VERI/O Laboratory Services Fit in the Drug Development Process
| Preclinical & Retrospective | Prospective Patient Enrollment | Companion Diagnostics (CDx) |
|---|---|---|
RUO |
IUO |
IVD |
| Analytical validation | Labeled & distributed under approved clinical protocol | Global commercialization Global regulatory approvals Clinical validation |
| VERI/O Laboratory Services or your lab | Qualified Service Provider (QSP) or Contract Research Organization (CRO) | Laboratories worldwide |
Benefits
- Molecular profiling expertise - highly trained and dedicated staff delivering high-quality data leveraging the advantages of the HTG EdgeSeq technology.
- Focused on turnaround times - receive a customized report on tens to thousands of biomarkers to meet your timelines.
- Deferred investment cost - all the advantages of targeted next-generation sequencing (NGS)-based results without the capital investments, training, and labor costs.
Available Gene Expression Panels
Products/Services Comparison
There are several ways to take advantage of the unique benefits of the HTG EdgeSeq technology.
| VERI/O Laboratory Services | In Your Lab Using The HTG EdgeSeq System | HTG Qualified Service Providers | |
|---|---|---|---|
| HTG Transcriptome Panel | Yes | Yes | Yes |
| HTG EdgeSeq Custom Assay | Yes | Yes | Yes |
| HTG EdgeSeq DLBCL Cell of Origin Assay | Yes | Yes | Yes |
| HTG EdgeSeq Immune Response Panel | Yes | Yes | Yes |
| HTG EdgeSeq miRNA Whole Transcriptome Assay | Yes | Yes | Yes |
| HTG EdgeSeq Mouse mRNA Tumor Response Panel | Yes | Yes | Yes |
| HTG EdgeSeq Oncology Biomarker Panel | Yes | Yes | Yes |
| HTG EdgeSeq Pan B-Cell Lymphoma Panel | Yes | Yes | Yes |
Applications in Immuno-Oncology
| Applications | Features | Benefits |
|---|---|---|
| Tumor microenvironment |
Measure the gene expression of microdissected tumor subsections such as vasculature, stroma, and tumor from a single FFPE tissue section |
Develop insights into the tumor biology
|
| Cytokine / chemokines |
Simultaneous monitoring of multiple chemokines and cytokines within cancer tissue and associated stroma |
Spatial-temporal mapping of immune-stimulatory or suppressant signals |
| Resistance / relapse system biology |
Characterization of pathway modulators that impact cancer drug response |
|
| Histology |
Measure the expression of cytokeratins, clusters of differentiation, epithelial genes and other histological markers |
|
| Signaling pathways |
Assess the status of pathways such as:
|
Grouping tumors via common dysregulated molecular pathways can provide deeper molecular understanding of tumors |
Other Applications
| Applications | Features | Benefits |
|---|---|---|
| Immune checkpoint expression within molecular subtyped diffuse large B-cell (DLBCL) tumors |
Characterization of immunocheckpoint factors together with tumor infiltrating lymphocyte (TIL) markers within DLBCL tumors classified via subtype |
Investigate tumor immune-responsiveness of DLBCL tumors |
| miRNA profiling of tumors and liquid biopsies |
Identify tumor miRNA-based signatures from different sample types using whole transcriptome miRNA profiling |
Identify tissue-based miRNA profiles that can be translated to blood samples |
| Immunophenotyping immune cells |
Pseudo-flow analysis using cluster of differentiation (CD)-based markers |
Sensitivity and ability to multiplex above applications/markers |
| Subtyping DLBCL tumors |
Characterize cases as Activated B-Cell Type (ABC) vs. Germinal Center B-Cell Type (GCB) |
Stratify groups for biomarker and drug development |
Sample Types and Input Requirements
Please contact your HTG Account Representative or support@htgmolecular.com for information regarding VERI/O sample input requirements
Learn More
For more information about VERI/O Laboratory Services, please fill out the form below. Or call us at (877) 507-3259.
Page last updated January 25, 2023
Ready to get started?
HTG is focused on delivering exceptional molecular profiling products and services. Contact us to find out how our unique technology can benefit you.